Summer Research Program for Science Teachers
Sausen Silmi, Canarsie HS, Brooklyn - 1997
Determining the Concentration
of a Solution: Beer's Law
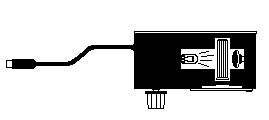
The primary objective of this experiment is to determine the concentration of an unknown nickel=(II) sulfate solution. You will be using the colorimeter shown in Figure 1. In this device, red light from the LED light source will pass through the solution and strike a photocell. The NiSO4 solution used in this experiment has a deep green color. A higher concentration of the colored solution absorbs more light (and transmits less) than a solution of lower concentration. [Content Standard B- Structure and properties of matter] The colorimeter monitors the light received by the photocell as either an absorbance or a percent transmittance value.

Figure 1
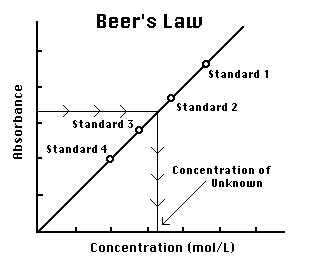
Figure 2
You are to prepare five nickel sulfate solutions of known concentration (standard solutions). Each is transferred to a small, rectangular cuvette that is placed into the colorimeter. The amount of light that penetrates the solution and strikes the photocell is used to compute the absorbance of each solution. When a graph of absorbance vs. concentration is plotted for the standard solutions, a direct relationship should result, as shown in Figure 2. The direct relationship between absorbance and concentration for a solution is known as Beer's law. [Content Standard Unifying Concepts- Change, constancy, and measurement]
The concentration of an unknown NiSO4 solution is then determined by measuring its absorbance with the colorimeter. By locating the absorbance of the unknown on the vertical axis of the graph, the corresponding concentration can be found on the horizontal axis (follow the arrows in Figure=2). The concentration of the unknown can also be found using the slope of the Beer's law curve.
MATERIALS [Teaching Standard D- Make accessible science tools and resources]
| CBL System |
30 mL of 0.40 M NiSO4 |
| TI Graphing Calculator | 5 mL of NiSO4 unknown solution |
| Vernier Colorimeter | two 10-mL pipets (or graduated cylinders) |
| Vernier adapter cable | pipet pump or pipet bulb |
| TI-Graph Link | distilled water |
| one cuvette | test tube rack |
| five 20 X 150 mm test tubes | two 100-mL beakers |
| tissues (preferably lint-free) | stirring rod |
PROCEDURE
1. Obtain and wear goggles! CAUTION: Be careful not to ingest any NiSO4 solution or spill any on your skin. Inform your teacher immediately in the event of an accident. [Teaching Standard D- Ensure a safe working environment]
2. Add about 30 mL of 0.40 M NiSO4 stock solution to a 100-mL beaker. Add about 30 mL of distilled water to another 100-mL beaker.
3. Label four clean, dry, test tubes 1-4 (the fifth solution is the beaker of 0.40 M NiSO4). Pipet 2, 4, 6, and 8 mL of 0.40 M NiSO4 solution into Test Tubes 1-4, respectively. With a second pipet, deliver 8, 6, 4, and 2 mL of distilled water into Test Tubes 1-4, respectively. Thoroughly mix each solution with a stirring rod. Clean and dry the stirring rod between stirrings. Keep the remaining 0.40 M NiSO4 in the 100-mL beaker to use in the fifth trial. Volumes and concentrations for the trials are summarized below:
| Trial Number |
0.40 M NiSO4 (mL) |
H2O (mL) |
Concentration (M) |
1 |
2 |
8 |
0.08 |
2 |
4 |
6 |
0.16 |
3 |
6 |
4 |
0.24 |
4 |
8 |
2 |
0.32 |
5 |
~10 |
0 |
0.40 |
4. Plug the colorimeter into the adapter cable in Channel 1 of the CBL System. Use the link cable to connect the CBL System to the TI Graphing Calculator. Firmly press in the cable ends. [Content Standard E- New technologies]
5. Turn on the CBL unit and the calculator. Start the CHEMBIO program and proceed to the MAIN MENU.
6. Set up the calculator and CBL for the colorimeter.
$ Select SET UP PROBES from the MAIN MENU.
$ Enter A1@ as the number of probes.
$ Select COLORIMETER from the SELECT PROBE menu.
$ Enter A1@ as the channel number.
7. You are now ready to calibrate the colorimeter. First prepare a blank by filling a cuvette 3/4 full with distilled water. To correctly use a colorimeter cuvette, remember:
$ All cuvettes should be wiped clean and dry on the outside with a tissue.
$ Handle cuvettes only by the top edge of the ribbed sides.
$ All solutions should be free of bubbles.
$ Always position the cuvette with its reference mark facing toward the white reference mark at the right of the cuvette slot on the colorimeter.
To calibrate the cuvette at 0% and 100% transmittance:
$ Place the blank cuvette in the cuvette slot of the colorimeter and close the lid. Turn the wavelength knob of the colorimeter to the 0% T position. In this position, the light source is turned off, so no light is received by the photocell. When the voltage reading displayed on the CBL screen stabilizes, press
on the CBL and enter A0@ (the Areference@) in the TI calculator.
$ Turn the wavelength knob of the colorimeter to the Red LED position (635 nm). In this position, the colorimeter is calibrated to show 100% of the red light being transmitted through the blank cuvette. When the displayed voltage reading stabilizes, press
and enter A100@ in the calculator. Leave the wavelength knob of the colorimeter set to the Red LED position for the remainder of the experiment.
$ Press
to return to the MAIN MENU.
8. Set up the calculator and CBL for data collection.
$ Select COLLECT DATA from the MAIN MENU.
$ Select TRIGGER/PROMPT from the DATA COLLECTION menu.
9. You are now ready to collect absorbance-concentration data for the five standard solutions. Empty the water from the cuvette. Using the solution in Test Tube 1, rinse the cuvette twice with ~1-mL amounts and then fill it 3/4 full. Wipe the outside with a tissue and place it in the colorimeter. After closing the lid, wait for the percent transmittance value displayed on the CBL screen to stabilize. Then press
and enter A0.080@ (the concentration, in mol/L) in the TI calculator. The absorbance and concentration values have now been saved for the first solution.
10. Select MORE DATA. Discard the cuvette contents as directed by your teacher. Rinse the cuvette twice with the Test Tube 2 solution, 0.16 M NiSO4, and fill the cuvette 3/4 full. Wipe the outside, place it in the colorimeter, and close the lid. When the percent transmittance value displayed on the CBL has stabilized, press
and enter A0.160@ in the calculator.
11. Repeat the Step 10 procedure to save the absorbance and concentration values of the solutions in Test Tube 3 (0.24 M) and Test Tube 4 (0.32 M), as well as the stock 0.40 M NiSO4. Wait until Step 14 to do the unknown.
12. Select STOP AND GRAPH from the DATA COLLECTION menu when you have finished collecting data. Examine the data points along the displayed graph of absorbance vs. concentration. As you move the cursor right or left, the concentration (X) and absorbance (Y) values of each data point are displayed below the graph. Record the absorbance values in your data table (round to the nearest 0.001).
13. To determine if your data is consistent with Beer>s law, plot a graph of absorbance vs. concentration with a linear regression curve displayed. If your data is consistent with Beer>s law (a direct relationship between absorbance and concentration), the regression line should closely fit the five data points and should pass through (or near) the origin of the graph.
$ Press
, then select NO to return to the MAIN MENU.
$ Select FIT CURVE from the MAIN MENU. Important: Do not select SET UP PROBES on the MAIN MENUÑdoing so will clear the data lists.
$ Select LINEAR L 1, L2 (LINEAR c 1, c 2 on the TI-92). The linear-regression statistics for these two lists are displayed for the equation in the form:
y = ax + b
where y is absorbance, x is concentration, a is the slope, and b is the y-intercept. Record the slope, a, in the data table. Note: One indicator of the quality of your data is the size of b. It=is a very small value if the regression line passes through or near the origin. The correlation coefficient, r, indicates how closely the data points match up (or fit) the regression lineÑa value of 1.00 indicates a nearly perfect fit.
$ To display the linear-regression curve on the graph of absorbance vs. concentration, press
, then select SCALE FROM 0 from the SCALE DATA menu. You can now observe how closely your data comes to a direct relationship (Beer>s law).
14. Obtain about 5 mL of the unknown NiSO4 in another clean, dry, test tube. Record the number of the unknown in the Data and Calculations table. Rinse the cuvette twice with the unknown solution and fill it about 3/4 full. Wipe the outside of the cuvette, place it into the colorimeter, and close the lid.
15. To find the absorbance of the unknown NiSO4 solution:
$ Press
to return to the MAIN MENU. Important: Do not select SET UP PROBES on the MAIN MENUÑdoing so will clear the data lists.
$ Select COLLECT DATA from the MAIN MENU.
$ Select MONITOR INPUT from the DATA COLLECTION MENU. Press
to monitor the colorimeter. The absorbance value of the unknown is displayed on the screen of the TI calculator. When the absorbance reading stabilizes, record its value in Trial 6 of the Data and Calculations table (round to the nearest 0.001).
$ Press
to quit MONITOR INPUT.
16. Discard the solutions as directed by your teacher. Proceed directly to Steps 1 and 2 of Processing the Data.
PROCESSING THE DATA
1. Determine the concentration of the unknown NiSO4 solution. Divide the absorbance value you obtained in Trial 6 by the slope of the regression line.
2. To confirm the unknown concentration value you obtained in Step 1, you can use your TI calculator and interpolate along the regression line on your Beer>s law curve. Use the following method to convert the absorbance value of the unknown to concentration, in mol/L.
$ Select FIT CURVE from the MAIN MENU.
$ Select LINEAR L 1, L2 (LINEAR c 1, c 2 on the TI-92).
$ To display the linear-regression curve on the graph of absorbance vs. concentration, press
, then select SCALE FROM 0 from the SCALE DATA menu.
$ To interpolate along the regression line, press
once (TI-82 or TI-83) or press the upper edge of the TI-92 cursor pad (not necessary on TI-85 calculators). Move the cursor left or right along the regression line. A cursor is displayed on the regression line, along with its X and Y coordinates below the graph. Move the cursor to an absorbance value (Y value) that is equal or nearly equal to the absorbance reading of your unknown sample. The concentration of the unknown (X value) will be approximately equal to the value you obtained in Step 1 above.
$ Use the TI-Graph Link cable and program to transfer the graph of absorbance vs. concentration, with a regression line and interpolated unknown concentration displayed, to a Macintosh or IBM-compatible computer. Print a copy of the graph.
DATA AND CALCULATIONS
| Trial |
Concentration (mol / L) |
Absorbance |
| 1 |
0.080 |
________= |
2 |
0.16 |
________= |
3 |
0.24 |
________= |
4 |
0.32 |
________= |
5 |
0.40 |
________= |
| 6 |
Unknown number |
_____= |
________= |
| Slope of regression line (m) |
________ L/mol |
| Concentration of the unknown |
________ mol/L |